This project was born from a basic concept. Surgeons we excited about understanding "soft-tissue", or how muscles were affected and could better recover after hip surgery and how they could better anticipate and avoid hip dislocation. We started out by asking our key thought leaders of what it was that mattered to them with user interviews.
Formus Navigator
Designing trusted AI decision support for surgical planning
Senior Product Designer
scope
End-to-end discovery, experience strategy, workflow design and validation
Strategic Contribution
Led user research, product strategy, go-to-market strategy, stakeholder alignment across R&D, engineering and clinical teams.
Product Context
AI-assisted surgical planning platform used by orthopaedic clinicions
1 year 7 months
Regulated Med-Tech | AI Software | B2B | SaaS
MMR
• Converted 12-month biomechanics research into live, mobile-responsive web app now used in real surgeries.
• Cut surgeon screening time from raw data tables to 30-second traffic-light scan.
• Converted sideline research project to second highest product priority.
• 5,000+ LinkedIn impressions
Project Overview
Navigator focused on translating emerging AI capabilities into clinically trusted decision workflows. The challenge was not only usability, but enabling surgeons to confidently rely on automated recommendations within high-risk treatment planning contexts. My role involved shaping product direction through research synthesis, defining interaction models that communicated system confidence, and accelerating implementation through engineering-aware prototyping. Working closely with researchers, engineers, surgeons, and commercial teams to translate complex bio-mechanical research into a fast, trusted experience surgeons could use in seconds.
• Medical device industry: $500B+ global market with strict regulatory requirements.
• Orthopedic surgery planning: critical need for dislocation risk assessment.
• AI in healthcare: Emerging technology requiring trust-building design.
• B2B medical SaaS: Complex sales cycles with multiple stakeholder groups.
• Collaborated with Head of Research, Biomechanics Specialist, AI Engineer, CEO.
• Guided technical team on user-centred design principles.
• Balanced clinical accuracy with commercial viability.
• Navigated regulatory considerations from medical software.
COnstraints
•Regulatory safety requirements in medical software (risk of misdiagnosis or clinician trust loss) required extreme caution on automation and messaging design.
• Limited engineering resources meant we could not build full automation initially, the project required staged manual validation (see rollout detail below).
Trade-offs
• Decided not to automate AI fully in beta opting for manual review + human-in-the-loop verification to ensure clinical accuracy and trust, delaying automation but reducing clinical risk.
Business Awareness
• Target metric: $2K MRR by end of 2025 tied directly to company OKRs. This influenced prioritisation of features (simple risk indicator over richer analytics).
• Adoption risk: surgeons are highly risk-averse and slow to adopt new tools. Early sign-ups driven by trust signals (KOL endorsements, clinical validation messaging).

some more
The scope of this project was over a year long and much of that time was spent on us working to understand and refine what surgeons wanted and how to best present that information to them.
Initial prototypes were confusing for surgeons, leading us to redesign towards clarity.
Once we knew we had a product surgeons wanted to use, I created an effective go-to-market strategy.
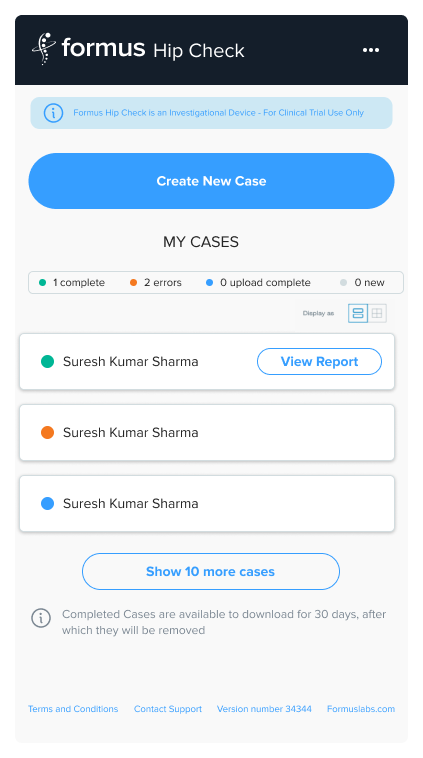
We used a range of tools and created a list of over 14 early surgeon sign ups, keen to test our new product. We eventually released it as a stand alone software product and is currently being used in real life surgeries with signed up, paying customers.
context & challenge
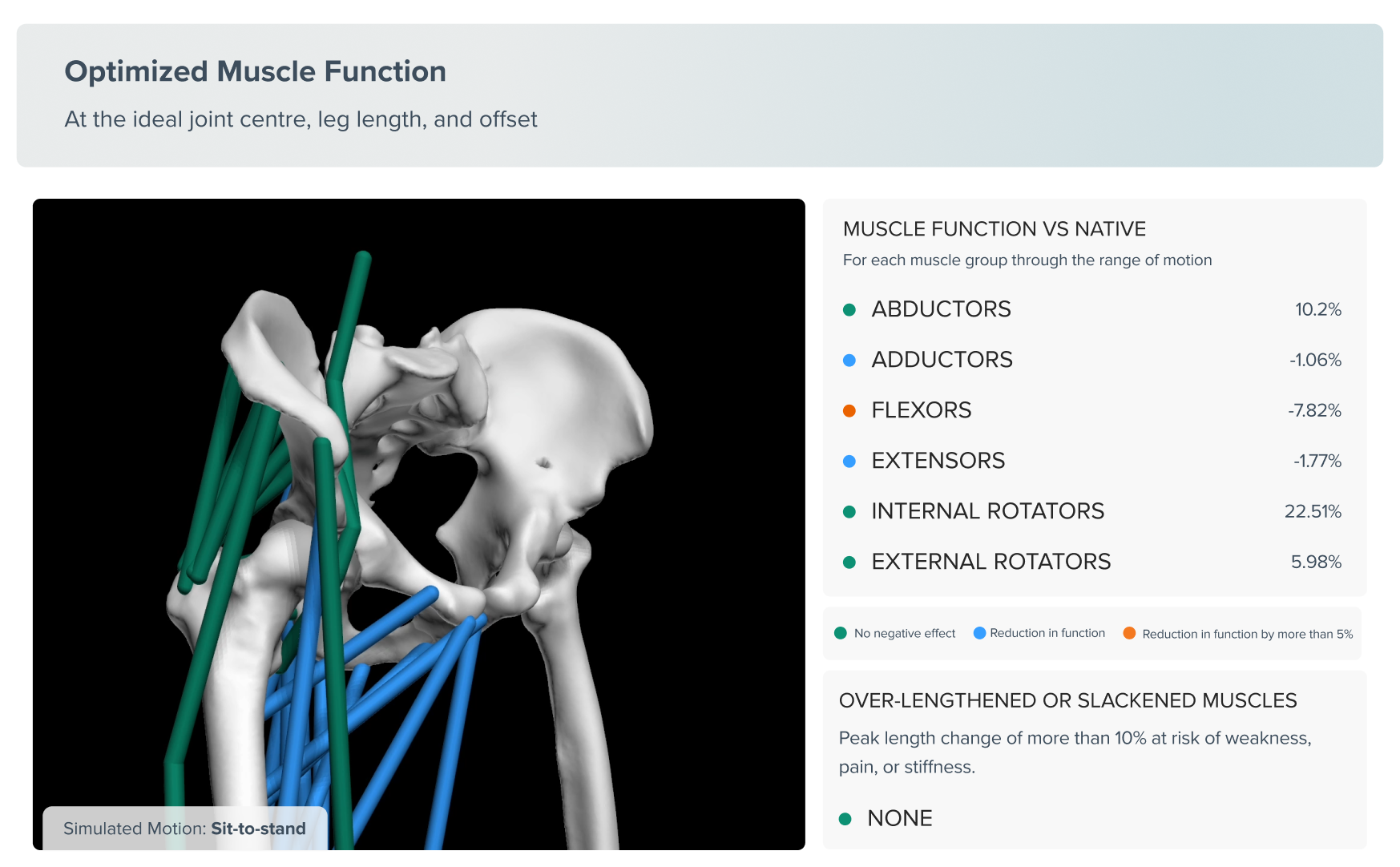
Formus was exploring how soft-tissue analysis and spinopelvic data could improve surgical outcomes, particularly around dislocation risk in hip replacement surgery. Early research produced valuable insights, but much of it lived in dense data tables and long-term concepts that were not yet commercially viable or practical for surgeons in day-to-day workflows.
The challenge was not simply visualising data, but identifying which problem to solve first. Surgeons needed something immediate, simple, and credible. Without that clarity, there was a risk of building an academically interesting tool that would struggle to gain real adoption.
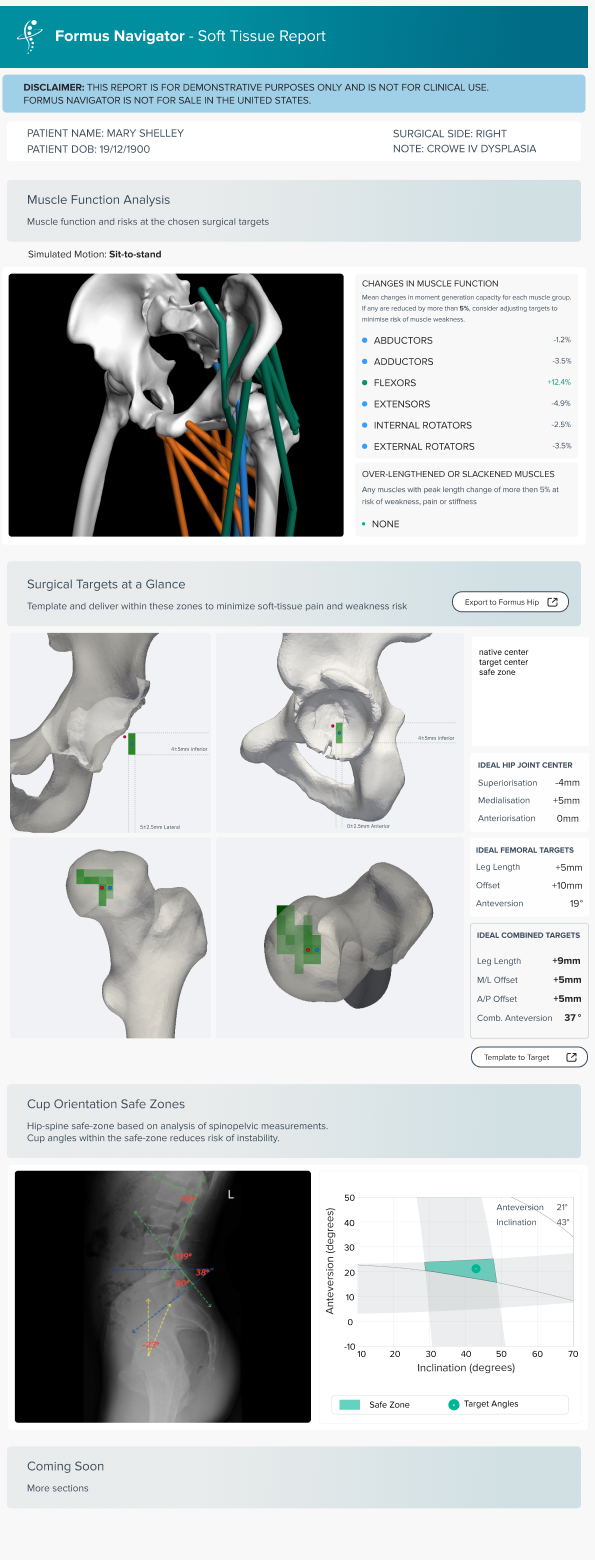
Surgical planning decisions carry irreversible patient outcomes. Introducing AI-generated recommendations into this workflow required careful design of transparency, confidence signalling, and cognitive load reduction.
The goal was to enable clinicians to interpret system outputs quickly while maintaining professional autonomy and trust in the planning process.
Discovery and problem framing
Initially product requirements were still emerging and clinical expectations varied across regions. I structured early discovery by synthesising stakeholder assumptions, reviewing prior research inputs, and identifying gaps in understanding around surgeon trust and decision validation behaviours. This enabled clearer prioritisation of experience risks and informed the interaction principles used throughout the design phase.
Discovery focused on understanding how surgeons currently assess dislocation risk, what signals they trust, and how much time they realistically have during planning. I led and supported interviews with surgeons, KOLs, and internal clinical experts, alongside ongoing feedback from sales and research teams.
A key insight emerged early. While soft-tissue modelling was compelling, surgeons were more concerned with quickly identifying high-risk patients. They needed a screening tool they could trust at a glance, not a deep analysis tool that required interpretation. This insight prompted a major pivot in product direction, from long-term exploration to solving an immediate, high-value clinical problem.
Discovery Trade-offs
Trade-off: Breadth vs Depth
After initial interviews, we debated collecting many shallow insights vs deep structured case studies. We chose deep interviews with a few Key Opinion Leaders (KOLs) to uncover actionable clinical needs, increasing confidence in product direction, even though it slowed early momentum.
Impact on Business
Deep insights ensured feature relevance (validated by surgeon prioritisation) and reduced later redesign cost; a strategic investment in long-term adoption (as reflected in early trial metrics).
Research and Design




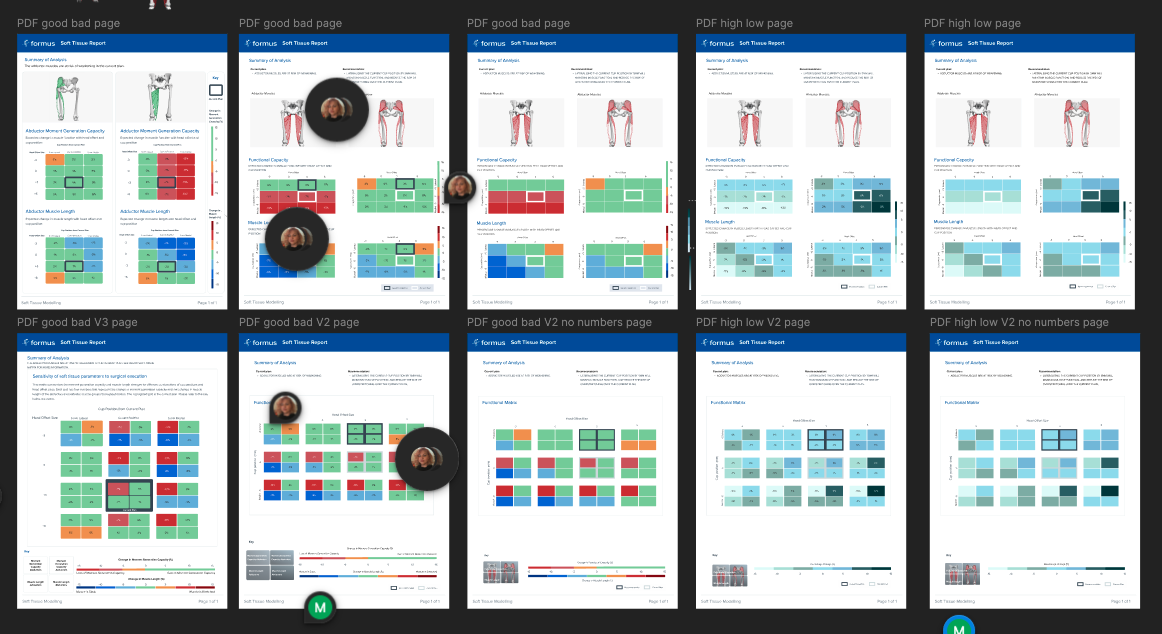








This shift reframed Navigator’s purpose. Instead of asking how much data we could show, the question became how little information we needed to confidently flag risk. The product moved toward answering a simple clinical question: is this patient at risk, and should I look closer?
This reframing aligned the product with real surgical workflows and laid the foundation for trust. It also helped align stakeholders across research, engineering, and product around a shared goal.
Iteration was continuous and research-led. Feedback loops with surgeons helped refine language, thresholds, and visual emphasis, ensuring the UI aligned with how risk is assessed clinically. The introduction of a traffic-light style system marked a turning point, allowing surgeons to scan results in seconds while still retaining access to deeper detail when needed.
Each design decision focused on balancing simplicity with credibility. The interface needed to feel clinically trustworthy, grounded in research, and defensible in a regulated environment.
A core focus was enabling clinicians to understand the intent and reliability of automated recommendations.
I explored interface patterns that communicated system certainty without overwhelming users with technical detail. This included progressive disclosure of supporting data, spatial visual cues reinforcing anatomical context, and interaction flows that allowed rapid comparison between manual and AI-assisted planning approaches.
Observability & UX Signals
Because Navigator’s AI outputs were complex and trusted, we defined UX signals to measure quality and risk:
• Error alerts: explicit doubt indicators when AI confidence was below threshold
• User override patterns: logs of when surgeons manually adjusted risk indicators
• Session drop-offs: where surgeons exited without completing a scan.
These signals informed prioritisation for design iteration and helped stakeholders quantify usability vs risk.
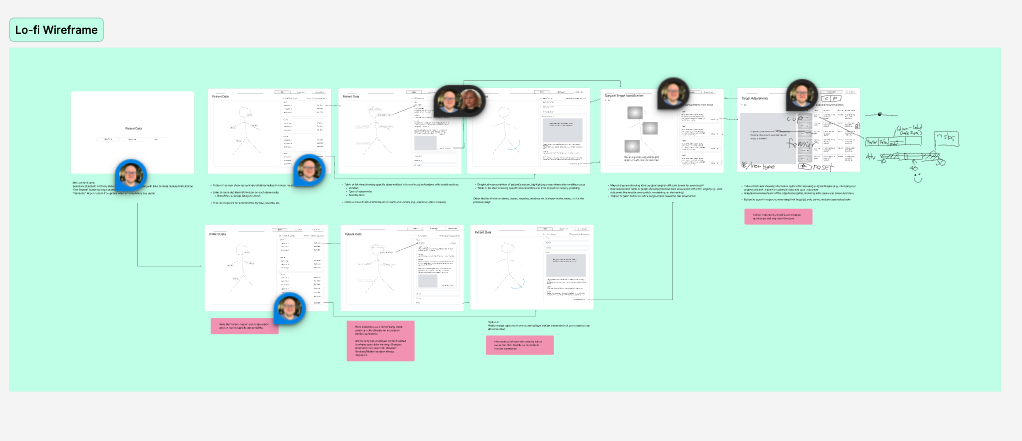
Wireframe Prototyping



With the problem clearly defined, I explored multiple ways to surface risk information, starting with low-fidelity wireframes and rapidly progressing to interactive prototypes. Early concepts focused on presenting raw measurements, but testing quickly showed that this increased cognitive load rather than reducing it.
Through iteration, the UI evolved from data tables to simplified visual cues, prioritising clarity, hierarchy, and speed. Prototypes were shared with surgeons and internal teams to validate comprehension and confidence before moving into higher-fidelity design.
One of the biggest challenges was the classic credibility gap of launching something new in a risk-averse clinical setting. This was addressed through a research-first beta approach, working closely with supportive KOLs and early adopters who were willing to trial Navigator in real cases.
This approach allowed us to gather real-world validation while remaining careful and ethical. It also demonstrated that even in regulated environments, it is possible to run lean, learn-as-you-go betas when trust and transparency are prioritised.
Navigator was designed as part of a broader surgical planning ecosystem. I considered how new interaction models would influence existing workflows, training expectations, and cross-team implementation dependencies. This helped reduce downstream engineering rework and supported smoother clinical onboarding when released.
Safe Rollout Strategy (Feature Flags & Phased Release)
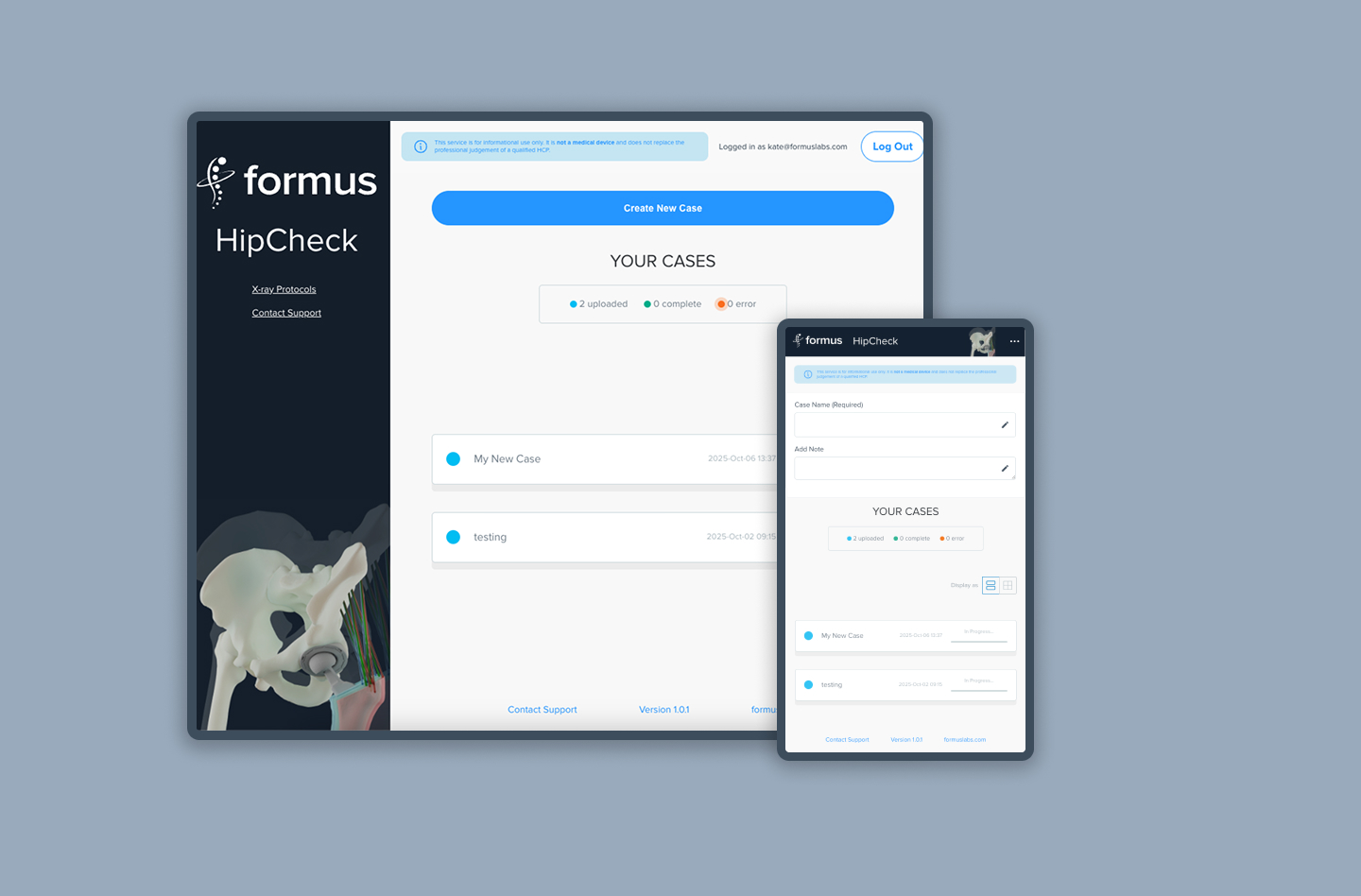
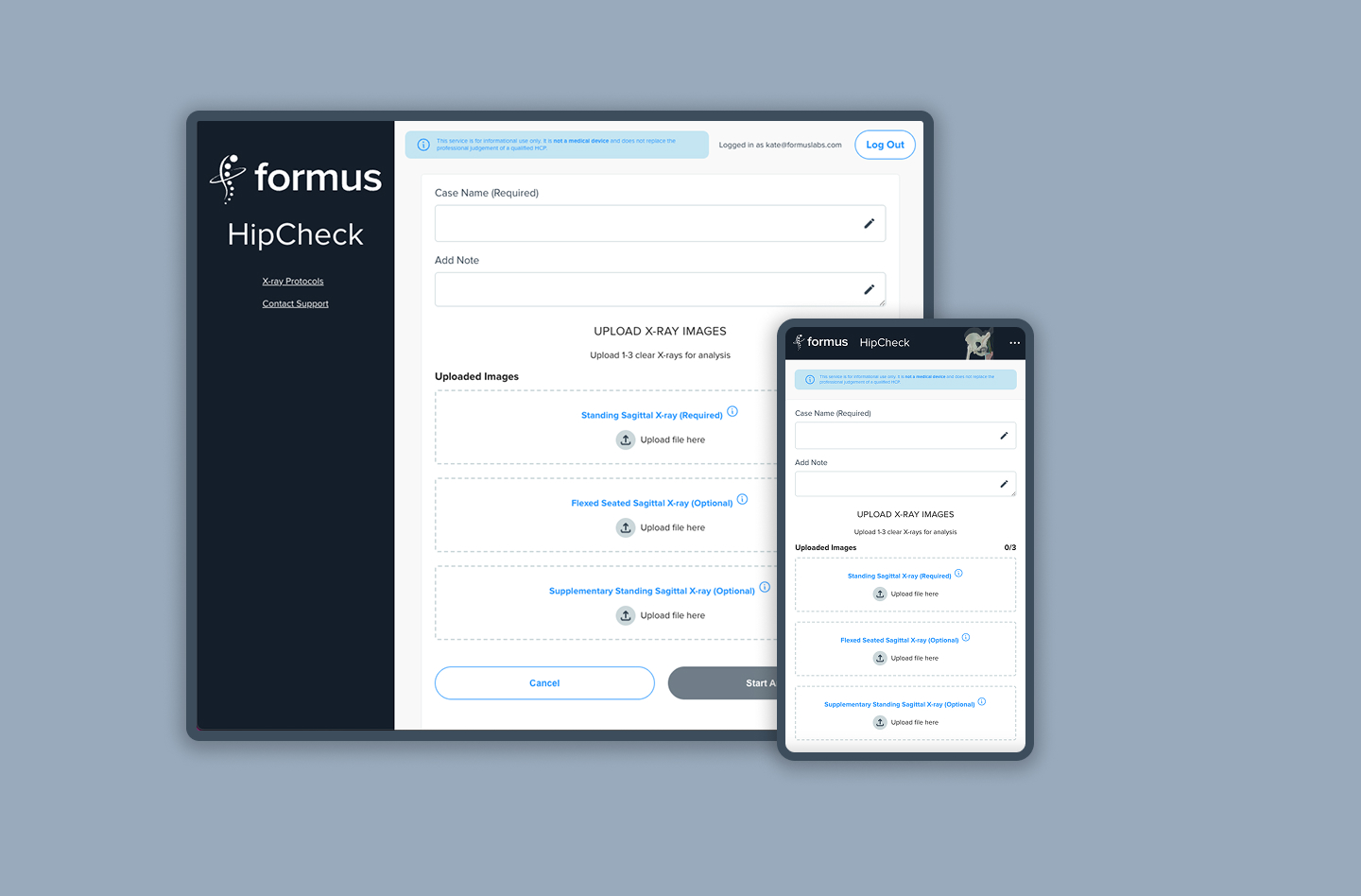
To reduce risk and gather real-world learning, we released a beta version behind a controlled access gate (effectively a feature flag/limited cohort rollout).
• Phase 1: Internal teams + selected surgeons:
Formus x-ray upload and manual data analysis: collect usability signals
• Phase 2: Stand-alone beta with opt-in surgeons:
User x-ray upload and manual data analysis: collect behavioural data and error patterns
• Phase 3: Expand to broader clinical partners once confidence thresholds met:
Integrate to Formus Hip Software under feature flag.
Trade-off
• Limited early access slowed full adoption but minimised risk of adverse clinical outcomes and gave structured data for iterative improvement.
CI/CD-Aware Design
• Although this was a Webflow beta, the conceptual feature gating and staged release aligns with CI/CD best practices, decoupling deployment from exposure and enabling controlled experimentation.
Go to market strategy
As confidence in the tool grew, Navigator shifted from a side project into a strategic product initiative. I contributed to shaping the go-to-market approach, aligning positioning, messaging, and early adoption strategies with the product’s core value: fast, trusted risk screening.
Alongside this, we worked towards defining next steps for growth, including identifying remaining knowledge gaps, uncovering friction in user flows, and planning further research to support broader adoption and integration into the Formus platform.
Navigator contributed to early market validation of the AI planning suite, supporting first revenue generation and positive clinician engagement during initial rollout. Research insights from the project also informed roadmap prioritisation across adjacent features with significant future commercial potential.
Revenue, Adoption & Market Constraints
Pricing experiment trade-off
We published pricing publicly to gauge market willingness, acknowledging internal hesitance to expose pricing before product maturity.
Adoption strategy
Balancing accessibility against clinical credibility, we chose LinkedIn outreach and KOL endorsements over broad paid campaigns to maximise quality of early trials with minimal spend.
Early sign-ups converting to paying customers validated willingness to pay before heavy investment in backend automation.
Customers and IMpact
Clinically, Navigator gives surgeons an at-a-glance way to screen for known predictors of dislocation, including stiff spine, flatback deformity, and posterior pelvic tilt. What began as dense research outputs became a practical tool that surgeons now trust to inform planning decisions.
From a product perspective, Navigator demonstrated how deep research could be translated into usable, scalable software. The UI became the unifying layer that connected research, product strategy, and commercial execution.
This project reinforced the importance of designing not only functional usability but psychological confidence when introducing AI into expert decision environments. By focusing on clarity of intent, progressive validation pathways, and engineering-aligned delivery practices, the team was able to move from conceptual capability to trusted clinical tool within a short timeframe.
Reflections and learnings
This project reinforced the importance of patience in discovery, particularly when working with highly specialised users.
Co-ordinating time with surgeons slowed early research, and in hindsight, formats like round-table discussions could have accelerated learning. A key lesson was knowing when to pivot from exploration to execution.
Soft-tissue research was valuable, but solving an immediate problem created momentum, trust, and adoption.
Using research synthesis, UX strategy, and go-to-market thinking to guide direction, I gained deep respect for the bio-mechanical engineers and researchers on the team. Where they worked in equations and models, my role was to translate that depth into clarity surgeons could act on. Navigator ultimately succeeded because rigorous research was paired with clear, human-centred design
Strategic Learnings
Business & Scale
• Next steps include integrating Navigator into wider product flows to improve stickiness and lifetime value. Investigating keeping it as a stand alone product and or as a new Formus Hip Integrated feature.
• Define agreed success thresholds (e.g. reduction in surgical complications or time saved) tied to commercial contracts, for both stand alone and Formus Hip integration.
AI Framing & Risks
• Fully automated AI screening was deprioritised in beta due to trust concerns and capacity. Future iterations will explore incremental automation only after clinical signal quality is measurable.
CI/CD Initiatives for Next Phase:
• Plan to work with engineering on true feature flags and environment staging to safely introduce new AI behaviours and automate deployments.
.gif)
.png)

